Flibanserin, the “female Viagra,” will be sold as Addyi. (Sprout Pharmaceuticals)
The first “female Viagra” pill has been approved for sale by the U.S. Food and Drug Administration. The agency on Tuesday approved Addyi, which will be sold by Sprout Pharmaceuticals. The drug is known generically as flibanserin.
“Today’s approval provides women distressed by their low sexual desire with an approved treatment option,” said Dr. Janet Woodcock, the director of the FDA’s Center for Drug Evaluation and Research. “The FDA strives to protect and advance the health of women, and we are committed to supporting the development of safe and effective treatments for female sexual dysfunction.”
The pill is designed to treat acquired, generalized hypoactive sexual desire disorder (HSDD) in premenopausal women, the FDA says. Its the first FDA-approved treatment for sexual desire disorders.
Female sexual dysfunction affects up to 1 in 10 women in the United States, according to Sprout Pharmaceuticals, the company that developed Addyi.
Here’s what you need to know:
1. The Pill Was Approved After 2 Previous Rejections
Flibanserin, developed for commercial sale as Addyi by Sprout Pharmaceuticals, was rejected twice by the FDA, in 2010 and 2013. This was the second time Sprout Pharmaceuticals led the fight for approval after the small company dedicated to women’s health acquired the rights to the drug.
“It has been a remarkable journey to get to this breakthrough moment. Today we celebrate what this approval means for all women who have long awaited a medical treatment option for this life impacting condition,” Cindy Whitehead, chief executive officer of Sprout, said in a statement. “We applaud the FDA for putting the patient voice at the center of the conversation and for focusing on scientific evidence.”
Flibanserin was studied in more than 11,000 women.
“The FDA approval of Addyi is largely based on three North American, Phase 3, 24-week, randomized double blind, placebo-controlled, studies of premenopausal women with HSDD,” Sprout Pharmaceuticals said in its press release. “For premenopausal women with HSDD, Addyi has demonstrated improvements in desire for sex, reducing distress from the loss of sexual desire and increasing the number of satisfying sexual events.”
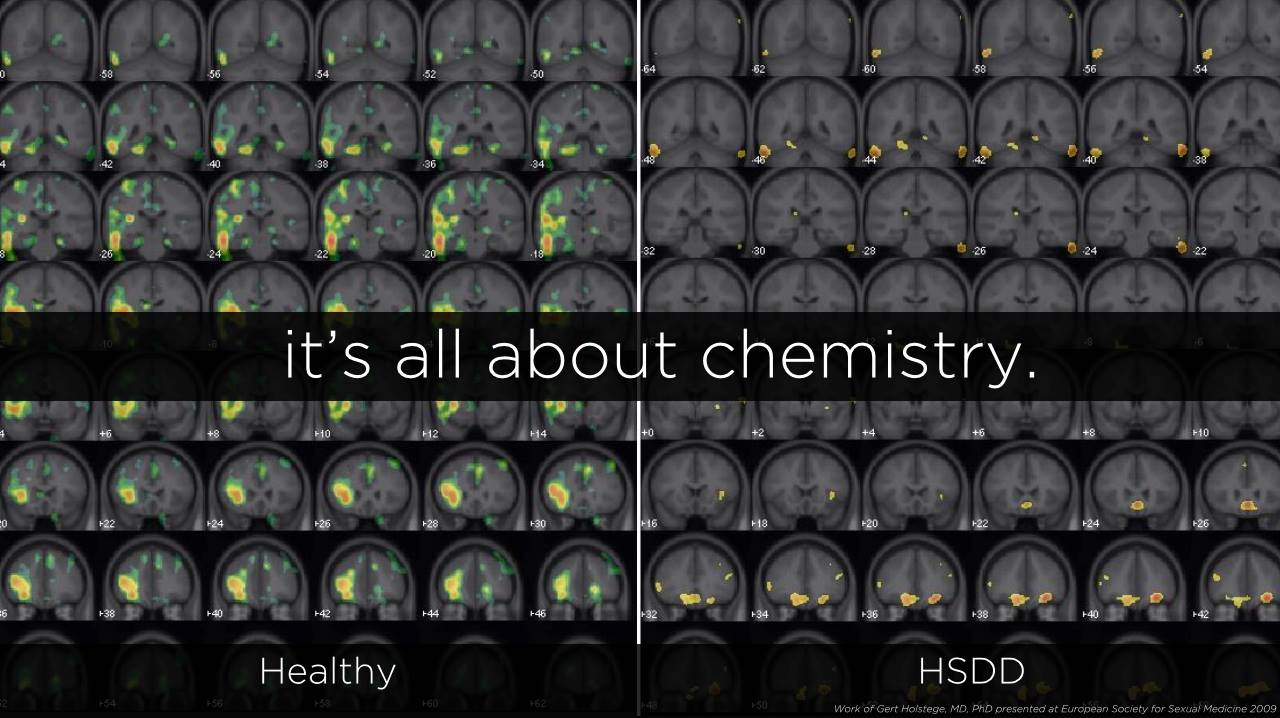
2. HSDD Causes a ‘Persistent Absence of Sexual Thoughts, Fantasies, Responsiveness & Willingness to Engage in Sexual Activity’
(Sprout Pharmaceuticals)
Sprout Pharmaceuticals said in its press release that hypoactive sexual desire disorder, or HSDD, “is defined as a persistent absence of sexual thoughts, fantasies, responsiveness and willingness to engage in sexual activity that causes personal or relationship distress and cannot be accounted for by another medical condition or substance. Acquired HSDD refers to HSDD that develops in a patient who previously had no problems with sexual desire. Generalized HSDD refers to HSDD that occurs regardless of the type of stimulation, situation or partner.”
The FDA says, “HSDD is characterized by low sexual desire that causes marked distress or interpersonal difficulty and is not due to a co-existing medical or psychiatric condition, problems within the relationship, or the effects of a medication or other drug substance. HSDD is acquired when it develops in a patient who previously had no problems with sexual desire. HSDD is generalized when it occurs regardless of the type of sexual activity, the situation or the sexual partner.”
3. The FDA-Approval Comes With a ‘Boxed Warning’
The FDA said in its press release that Addyi received approval with a “Boxed Warning” to “highlight the risks of severe hypotension and syncope in patients who drink alcohol during treatment with Addyi, in those who also use moderate or strong CYP3A4 inhibitors, and in those who have liver impairment. Addyi is contraindicated in these patients.”
It is requiring Sprout Pharmaceuticals to “conduct three well-designed studies in women to better understand the known serious risks of the interaction between Addyi and alcohol.”
The warning addresses some concern about the safety of the drug, which had halted its approval in the past and drew opposition to it.
Other adverse reactions include, “dizziness, somnolence (sleepiness), nausea, fatigue, insomnia and dry mouth,” according to the FDA release.
“Because of a potentially serious interaction with alcohol, treatment with Addyi will only be available through certified health care professionals and certified pharmacies,” said Dr. Janet Woodcock, the director of the FDA’s Center for Drug Evaluation and Research. “Patients and prescribers should fully understand the risks associated with the use of Addyi before considering treatment.”
4. Sprout Pharmaceuticals Is a North Carolina-Based Company Founded to Focus on Flibanserin

The Sprout Pharmaceuticals staff, led by co-founder and CEO Cindy Whitehead. (SproutPharmaceuticals.com)
Sprout Pharmaceuticals, a small, private company, was founded with a focus on gaining approval for flibanserin and selling it commercially as Addyi. According to BizJournals.com, the Raleigh, North Carolina-based company raised a $100 million war chest in anticipation of approval.
“Singular in our focus, our leadership team sold off a successful business in male sexual health to take on the pursuit of this much needed scientific and societal breakthrough for women,” Sprout says in its mission statement.
CEO Cindy Whitehead and the other founders came from Slate Pharmaceuticals, which developed Testopel, the first FDA-approved “long-acting testosterone preparation for men.”
Whitehead, “led the sale of the Slate to GTCR/Actient Pharmaceuticals in December 2011 spinning out Sprout Pharmaceuticals to pursue the first ever FDA drug approval for women’s most common sexual dysfunction,” according to Sprout’s website.
5. The Pill Is Expected To Be Available in October
According to Sprout, the pill is anticipated to be available by October 17, 2015.
Updates on the availability of Addyi are available on the website Sprout set up for the pill.
