
Getty A new Quest Diagnostics antibody test for COVID-19 is now available online.
You can purchase a new Quest Diagnostics antibody test for COVID-19 online. However, you have to physically go into one of the company’s locations to get your blood drawn in order for it to work.
The goal of antibody tests is to determine whether a person’s body has produced antibodies as a result of a recovered coronavirus infection, perhaps giving them some level of immunity to the virus. However, be aware that researchers are still trying to understand exactly how much immunity – if any – people get once they’ve recovered from COVID-19. The World Health Organization has advised that such antibody claims are not proven and remain under study. You can read more about that advisement in detail toward the end of this article.
Heavy went on the Quest Diagnostics site and went through the process of purchasing the online test, putting in this author’s hometown in the metropolitan area of Milwaukee. The site, on April 28, said the blood test was available near a local mall on the outskirts of Milwaukee on May 1. We also put in a rural address in Wisconsin, by way of another example, and were told there wasn’t a blood test available for 30 days, so it can vary depending on where you live. You would put your own city and state into the site to determine the availability and location of the blood draws in your area.
“This test checks for a type of antibody called immunoglobulin G (IgG) that is the result of past or recent exposure to COVID-19, also known as the novel coronavirus,” Quest Diagnostics explains. “The human body produces IgG antibodies as part of the immune response to the virus. It usually takes around 10 to 18 days to produce enough antibodies to be detected in the blood.”

What you see when you purchase a Quest Diagnostics antibody test online.
To get to the point where you can purchase the test, click here, and choose the $119 COVID-19 antibody test by clicking “add to cart.” You will be asked a series of questions: Your date of birth, which state you live in, whether you have “experienced fever higher than 100.5 °F in the last 3 days,” and whether you’ve experienced any of the “following new onset symptoms in the past 10 days”:
You will also be asked: Have you been tested for and diagnosed with COVID-19 infection? Have you been exposed to anyone with known COVID-19 infection in the last 14 days?
We answered that we were symptom free, weren’t exposed, and weren’t diagnosed with COVID-19. The site is trying to determine whether you have an active infection because, if so, you should be trying to get a test for COVID-19 (and contact your doctor), rather than a test for its antibodies, which can, as noted, take days to develop.
The item will then be added to your cart if you pass that step. When you click checkout, it will ask you to sign in as a new customer or create an account. We chose new customer. It then asks you to put in your city and state to schedule an appointment for the blood draw. We did this again as a test on April 29, and, one day after we first tried, the May 1 appointments were gone and the next one offered was 48 miles away in a different community on May 5, at least in our area, showing the popularity of the antibody test.
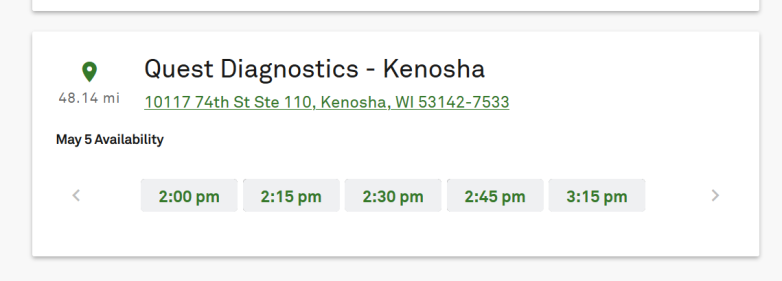
Quest DiagnosticsQuest Diagnostics appointment example
Again, this will vary depending on where you live and what symptoms you report.
Here’s what you need to know:
The Tests Aren’t for Active Infections
GettyThis photo from the Centers for Disease Control and Prevention (CDC) shows a microscopic view of the Coronavirus.
Quest isn’t offering tests for active infections. “COVID-19 active infection testing is not currently available through QuestDirect, and we are not collecting specimens in our Patient Service Centers,” the site explains.
“Please contact your healthcare provider if you think you are infected. Safety measures at our Patient Service Centers have been enhanced, including a requirement that all patients wear a mask or other face covering.”
What is the purpose of the antibody test? Quest explains:
Test results may help identify if you were previously exposed to the virus and, if exposed, can check whether or not your body has produced antibodies. Antibodies typically suggest protective immunity after you’ve recovered or been exposed to COVID-19. However, evidence is still being collected to determine if IgG antibodies provide protective immunity against SARS-CoV-2, the virus that causes COVID-19 infection.
The site argues that the test is valuable:
If you were never diagnosed with COVID-19, this test can help determine if you may have been previously exposed to the virus.
If you were diagnosed with COVID-19, this test can check whether or not your body has produced antibodies.
You can learn more about the Quest Diagnostics test here, including other limitations or warnings associated with it.
According to CBS News, Quest has 2,200 blood draw centers throughout the United States, and it takes one to two days to get the results.
The Science on Antibody Tests Is Still Evolving & Immunity Uncertain

A man wearing a protective mask talks with a healthcare worker to be tested for the coronavirus (COVID-19) outside Gouverneur Health Center on April 24, 2020, in New York City.
Do antibody tests even work? Scientists are raising caution because the COVID-19 virus is so new and still being understood. The World Health Organization has an entire page devoted to its stance on so-called immunity passports, which some people think should be given to people with antibodies so they could re-enter society and the workforce.
WHO reports:
The development of immunity to a pathogen through natural infection is a multi-step process that typically takes place over 1-2 weeks. The body responds to a viral infection immediately with a non-specific innate response in which macrophages, neutrophils, and dendritic cells slow the progress of virus and may even prevent it from causing symptoms. This non-specific response is followed by an adaptive response where the body makes antibodies that specifically bind to the virus. These antibodies are proteins called immunoglobulins. The body also makes T-cells that recognize and eliminate other cells infected with the virus. This is called cellular immunity. This combined adaptive response may clear the virus from the body, and if the response is strong enough, may prevent progression to severe illness or re-infection by the same virus. This process is often measured by the presence of antibodies in blood.
However, WHO wrote that it’s still reviewing the evidence on COVID-19 antibody response, adding that most studies do show that people who recovered have antibodies. However, some people have “very low levels” of antibodies in their blood, according to WHO.
“As of 24 April 2020, no study has evaluated whether the presence of antibodies to SARS-CoV-2 confers immunity to subsequent infection by this virus in humans,” WHO warned. “Laboratory tests that detect antibodies to SARS-CoV-2 in people, including rapid immunodiagnostic tests, need further validation to determine their accuracy and reliability.”
The WHO also indicated that some tests can’t tell the difference between antibodies produced by COVID-19 or other coronaviruses and can generate false positives or negatives.
The WHO wrote that it supports antibody studies but added:
At this point in the pandemic, there is not enough evidence about the effectiveness of antibody-mediated immunity to guarantee the accuracy of an ‘immunity passport’ or ‘risk-free certificate.’ People who assume that they are immune to a second infection because they have received a positive test result may ignore public health advice. The use of such certificates may therefore increase the risks of continued transmission. As new evidence becomes available, WHO will update this scientific brief.
READ NEXT: The Real Story of Waco.